At its simplest, a phenolic resin is the reaction product of a phenol and an aldehyde. The typical phenol used for the reaction is phenol itself, although alkylated phenols are also sometimes used to impart specific characteristics to the reaction product. The dominant aldehyde (>95%) used for resin production is formaldehyde, although this may be used in several forms ranging from the solid concentrate known as para-formaldehyde or ‘paraform’ to a variety of solutions in water which are usually referred to as ‘formalin’. The stability of formaldehyde solutions in water is such that some stabiliser is usually required. Methanol is traditionally used for this purpose, but the level required depends on the concentration of formalin under consideration. Typical formalin concentrations will vary between 37% and 55% with the more recent trend being for higher concentrations to save on the energy and time required to strip of the water during later processing.
At its simplest, a phenolic resin is the reaction product of a phenol and an aldehyde. The typical phenol used for the reaction is phenol itself, although alkylated phenols are also sometimes used to impart specific characteristics to the reaction product. The dominant aldehyde (>95%) used for resin production is formaldehyde, although this may be used in several forms ranging from the solid concentrate known as para-formaldehyde or ‘paraform’ to a variety of solutions in water which are usually referred to as ‘formalin’. The stability of formaldehyde solutions in water is such that some stabiliser is usually required. Methanol is traditionally used for this purpose, but the level required depends on the concentration of formalin under consideration. Typical formalin concentrations will vary between 37% and 55% with the more recent trend being for higher concentrations to save on the energy and time required to strip of the water during later processing.
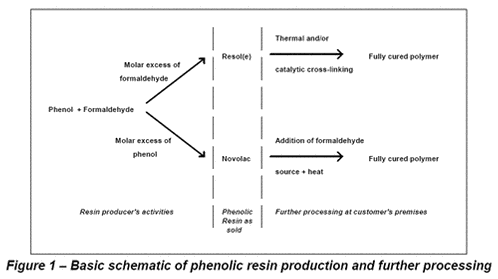
There are two types of phenolic resin that can be produced from phenol and formaldehyde. The first is produced when there is a molar excess of formaldehyde present and is known as a resol or resole. The second is produced when there is a molar excess of phenol present and is known as a novolac. In general, resoles tend to be liquids and novolacs tend to be solids. However, there are always exceptions to ‘prove the rule’. The solid nature of most novolacs requires them to be ground, flaked or pelletised before sale to the user. In some cases, novolacs are dissolved in solvent and sold as solutions. Perhaps the most important distinction between resoles and novolacs is that novolacs require the addition of a further formaldehyde source before full cross-linking can take place. Figure 1 illustrates the basic schematic flow.